
APA Style
Ken-ichiro Kamei, Batool Abedrabbo, Noor Alhaddad, Jody Aly, Juan Fernando Castaño, Zhansaya Makhambetova. (2026). Synthetic Polymer Based Nano Drug Delivery Systems for Precise Alzheimer’s Disease Therapy. Cell Therapy & Engineering Connect, 2 (Article ID: 0010). https://doi.org/Registering DOIMLA Style
Ken-ichiro Kamei, Batool Abedrabbo, Noor Alhaddad, Jody Aly, Juan Fernando Castaño, Zhansaya Makhambetova. "Synthetic Polymer Based Nano Drug Delivery Systems for Precise Alzheimer’s Disease Therapy". Cell Therapy & Engineering Connect, vol. 2, 2026, Article ID: 0010, https://doi.org/Registering DOI.Chicago Style
Ken-ichiro Kamei, Batool Abedrabbo, Noor Alhaddad, Jody Aly, Juan Fernando Castaño, Zhansaya Makhambetova. 2026. "Synthetic Polymer Based Nano Drug Delivery Systems for Precise Alzheimer’s Disease Therapy." Cell Therapy & Engineering Connect 2 (2026): 0010. https://doi.org/Registering DOI.
 ACCESS
Review Article
ACCESS
Review Article
Volume 2, Article ID: 2026.0010

Ken-ichiro Kamei
kk4801@nyu.edu

Batool Abedrabbo
ba2255@nyu.edu

Noor Alhaddad
nta4054@nyu.edu

Jody Aly
ja4534@nyu.edu

Juan Fernando Castaño
jfc9577@nyu.edu

Zhansaya Makhambetova
zm2285@nyu.edu
1 Nanobiotechnology Class, Program of Bioengineering, Division of Engineering, New York University Abu Dhabi, UAE
2 Program of Biology, Division of Science, New York University Abu Dhabi, Abu Dhabi, UAE
3 Department of Biomedical Engineering, Tandon School of Engineering, New York University, Brooklyn, NY 11201, USA
4 Department of Biology, Faculty of Arts & Science, New York University, NY, USA
5 Institute for Integrated Cell-Material Sciences (WPI-iCeMS), Institute for Advanced Study, Kyoto University; Yoshida-Ushinomiya-cho, Sakyo-ku, Kyoto, 606-8501, Japan
* Author to whom correspondence should be addressed
Received: 27 Sep 2025 Accepted: 20 Apr 2026 Available Online: 20 Apr 2026
Abstract:
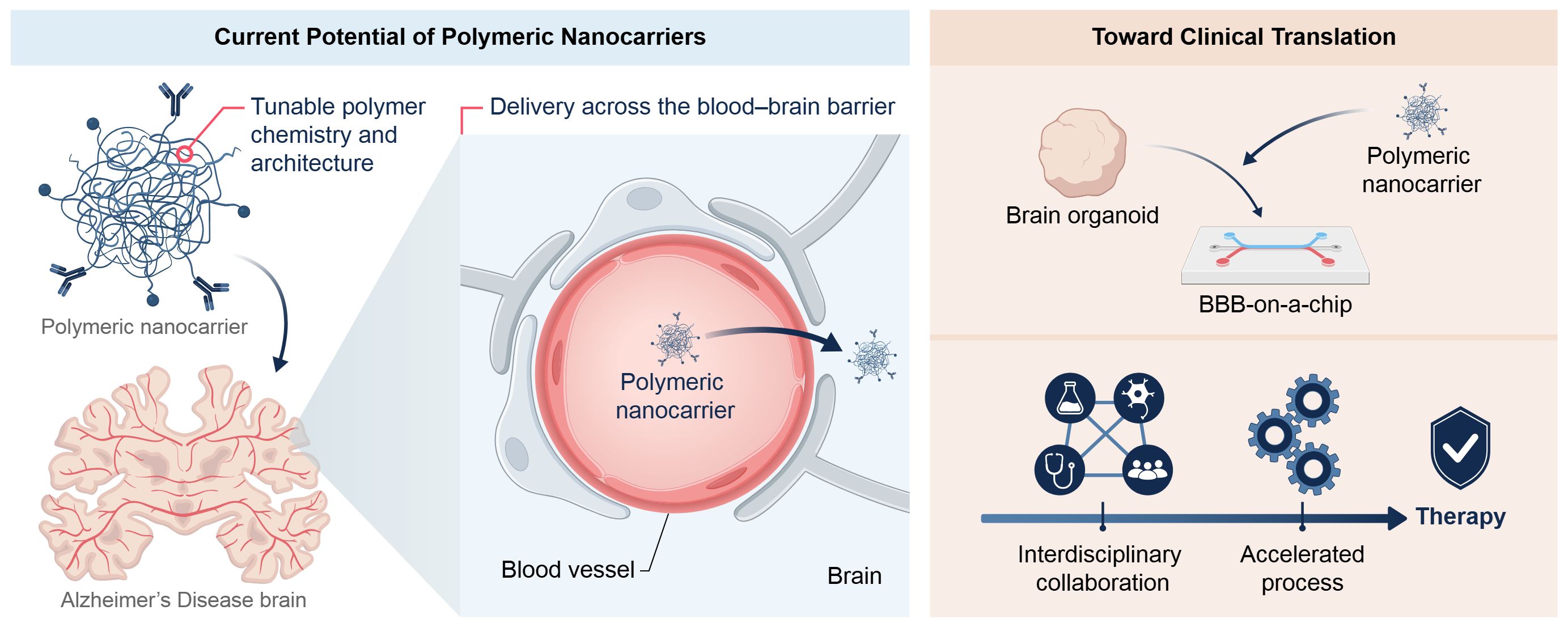
Alzheimer’s disease (AD) remains a major global health challenge due to its multifactorial pathology and the limited efficacy of existing therapies, which largely provide symptomatic relief without altering disease progression. A central obstacle to effective treatment is the blood–brain barrier (BBB), which severely restricts the delivery of therapeutics to the central nervous system. In this context, synthetic polymer–based nanoparticles have emerged as versatile platforms capable of improving drug stability, prolonging systemic circulation, and enabling controlled and targeted delivery across the BBB. This review summarizes recent advances in polymeric nanocarriers for AD therapy, with emphasis on translationally relevant systems, including poly(lactic-co-glycolic acid) (PLGA), poly(ε-caprolactone) (PCL), poly(lactic acid)–poly(ethylene glycol) (PLA–PEG), and polyethyleneimine (PEI). We discuss how polymer chemistry, degradation kinetics, and surface functionalization can be rationally engineered to overcome key delivery barriers, while critically addressing inherent trade-offs such as long-term biodegradability and clearance (PCL) and the balance between cytotoxicity and transfection efficiency (PEI). Emerging polymeric architectures, including dendrimers and hybrid systems, are also highlighted for their potential to support multifunctional and tunable delivery strategies. Importantly, we emphasize the increasing role of advanced human-relevant in vitro models—such as brain organoids and BBB-on-a-chip platforms—supported by recent regulatory initiatives promoting New Approach Methodologies (NAMs). These systems provide more predictive tools for evaluating nanoparticle transport, safety, and therapeutic response, thereby strengthening translational confidence. Collectively, this review argues that successful clinical translation of polymer-based nanotherapies for AD will depend on the development of hybrid polymer systems validated in advanced human models, alongside early consideration of manufacturability and regulatory alignment.
Disclaimer: This is not the final version of the article. Changes may occur when the manuscript is published in its final format.
We use cookies to improve your experience on our site. By continuing to use our site, you accept our use of cookies. Learn more