
APA Style
Avinash Kumar Singh, Roshni Sharma, Vidya Spriha Kujur, Mrinal Poddar, Ashish Kumar, Sanoj Kumar, Tarun Kumar Dhiman, Rahul Kumar. (2025). Integration of Nanotechnology and Nanomaterials in Biomaterials Research. Biomaterials Connect, 2 (Article ID: 0019). https://doi.org/Registering DOIMLA Style
Avinash Kumar Singh, Roshni Sharma, Vidya Spriha Kujur, Mrinal Poddar, Ashish Kumar, Sanoj Kumar, Tarun Kumar Dhiman, Rahul Kumar. "Integration of Nanotechnology and Nanomaterials in Biomaterials Research". Biomaterials Connect, vol. 2, 2025, Article ID: 0019, https://doi.org/Registering DOI.Chicago Style
Avinash Kumar Singh, Roshni Sharma, Vidya Spriha Kujur, Mrinal Poddar, Ashish Kumar, Sanoj Kumar, Tarun Kumar Dhiman, Rahul Kumar. 2025. "Integration of Nanotechnology and Nanomaterials in Biomaterials Research." Biomaterials Connect 2 (2025): 0019. https://doi.org/Registering DOI.
 ACCESS
Review Article
ACCESS
Review Article
Volume 2, Article ID: 2025.0019

Avinash Kumar Singh
avinashkumarsingh92@gmail.com

Roshni Sharma
roshnisharma2608@gmail.com

Vidya Spriha Kujur
vidyasphk94@gmail.com

Mrinal Poddar
mrinalpoddar02@gmail.com

Ashish Kumar
ashish.nano2011@gmail.com

Sanoj Kumar
sanoj156@gmail.com

Tarun Kumar Dhiman
tkdhiman91@gmail.com

Rahul Kumar
vns.rahul92@gmail.com
1 Foundation of research and technology- Hellas (FORTH), Heraklion, Crete, 70013, Greece
2 Department of Chemistry, Aligarh Muslim University, Aligarh, 202002, India
3 Center for Nanotechnology, Central University of Jharkhand, Ranchi, Jharkhand, 835205, India
4 Department of Chemical and Materials Engineering, Chang Gung University, Taoyuan 333, Taiwan
5 Centre for Studies in Science Policy, Jawaharlal Nehru University New Delhi, 110067, India
6 University School of Basic and Applied Sciences, Guru Gobind Singh Indraprastha University, New Delhi, 110078, India
* Author to whom correspondence should be addressed
Received: 12 Mar 2025 Accepted: 16 Aug 2025 Available Online: 24 Aug 2025
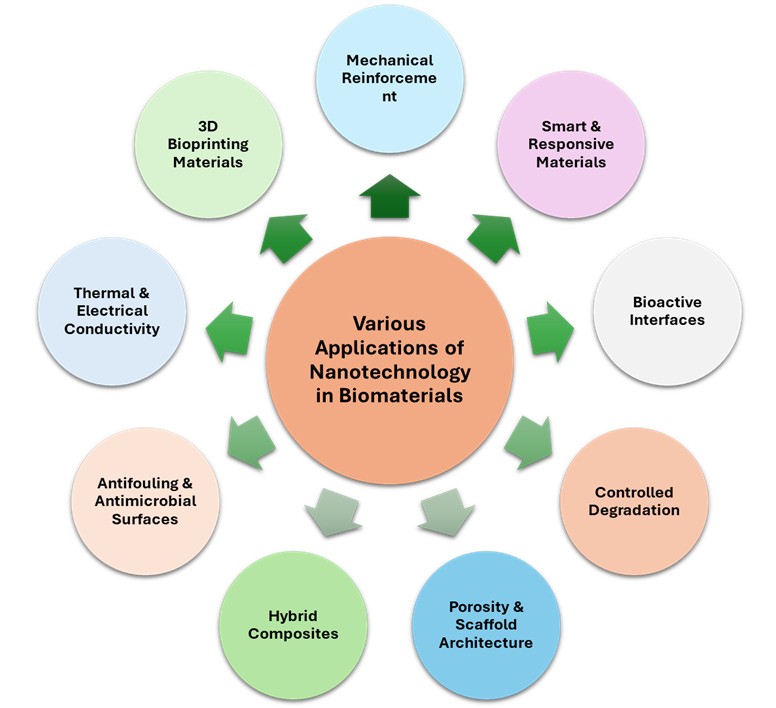
Nanotechnology has emerged as a revolutionizing element in biomaterials research, significantly enhancing their functionality and versatility in medical applications from tissue engineering, drug delivery, regenerative medicine, to medical implants. Integration of nanomaterials in biomaterials has led to an enormous enhancement in biocompatibility, mechanical strength, drug release control, and bioactivity. The present review provides an exhaustive overview of the historical perspective, classification, and applications of nanomaterials in biomaterials research. It talks about how inorganic, organic, and hybrid nanomaterials are contributing to advancing biomedical applications, including their impact on scaffolds, nanoparticles for targeted drug delivery, and surface modification for implants. The paper also considers the current challenges associated with the use of nanomaterials, including biocompatibility, toxicity, scalability, and regulation. Finally, future research directions are proposed to drive the safety, functionality, and integration of nanotechnology in biomaterials, with possibilities for next-generation biomedical applications. This review aims to highlight the profound influence of nanotechnology on biomaterials and its potential to revolutionize healthcare. It explores the transformative impact of nanomaterials on biological applications and focuses on specific applications such as tissue engineering, drug delivery systems, diagnostic instruments, and regenerative medicine.
Disclaimer : This is not the final version of the article. Changes may occur when the manuscript is published in its final format.
We use cookies to improve your experience on our site. By continuing to use our site, you accept our use of cookies. Learn more