
APA Style
Cletus Anes Ukwubile, Muhammad Zannah Lawan, Troy Salvia Malgwi, Hassan Braimah Yesufu. (2025). Evaluation of Anti-inflammatory and Anticancer Activities of Maerua angolensis DC. Leaf Extract-loaded Chitosan Nanoparticles. Biomaterials Connect, 2 (Article ID: 0018). https://doi.org/Registering DOIMLA Style
Cletus Anes Ukwubile, Muhammad Zannah Lawan, Troy Salvia Malgwi, Hassan Braimah Yesufu. "Evaluation of Anti-inflammatory and Anticancer Activities of Maerua angolensis DC. Leaf Extract-loaded Chitosan Nanoparticles". Biomaterials Connect, vol. 2, 2025, Article ID: 0018, https://doi.org/Registering DOI.Chicago Style
Cletus Anes Ukwubile, Muhammad Zannah Lawan, Troy Salvia Malgwi, Hassan Braimah Yesufu. 2025. "Evaluation of Anti-inflammatory and Anticancer Activities of Maerua angolensis DC. Leaf Extract-loaded Chitosan Nanoparticles." Biomaterials Connect 2 (2025): 0018. https://doi.org/Registering DOI.
 ACCESS
Research Article
ACCESS
Research Article
Volume 2, Article ID: 2025.0018

Cletus Anes Ukwubile
caukwubile@unimaid.edu.ng

Muhammad Zannah Lawan
mz4chem@unimaid.edu.ng

Troy Salvia Malgwi
troysm2013@unimaid.edu.ng

Hassan Braimah Yesufu
hbyesufu@unimaid.edu.ng
1 Department of Pharmacognosy, Faculty of Pharmacy, University of Maiduguri, PMB 1069, Maiduguri, Borno State, Nigeria.
2 Department of Chemical Engineering, Faculty of Engineering, University of Maiduguri, PMB 1069, Maiduguri, Borno State, Nigeria.
3 Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Maiduguri, PMB 1069, Maiduguri, Borno State, Nigeria.
* Author to whom correspondence should be addressed
Received: 07 Feb 2025 Accepted: 05 Aug 2025 Available Online: 20 Aug 2025
Background: Despite advancements in medicine, cancer and inflammation remain pressing issues of concern due to the negative impact of conventional treatment and the rising number of patients suffering from cancer. Medicinal plants' bioactive compounds have emerged as promising alternatives with Maerua angolensis gaining attention in Nigeria for its possible anti-cancer activity while being traditionally used for its anti-inflammatory properties.
Aim: The study is designed to evaluate the anti-inflammatory and anti-cancer effects of the extract of leaves of M. angolensis incorporated into chitosan nanoparticles (MALC-NPs).
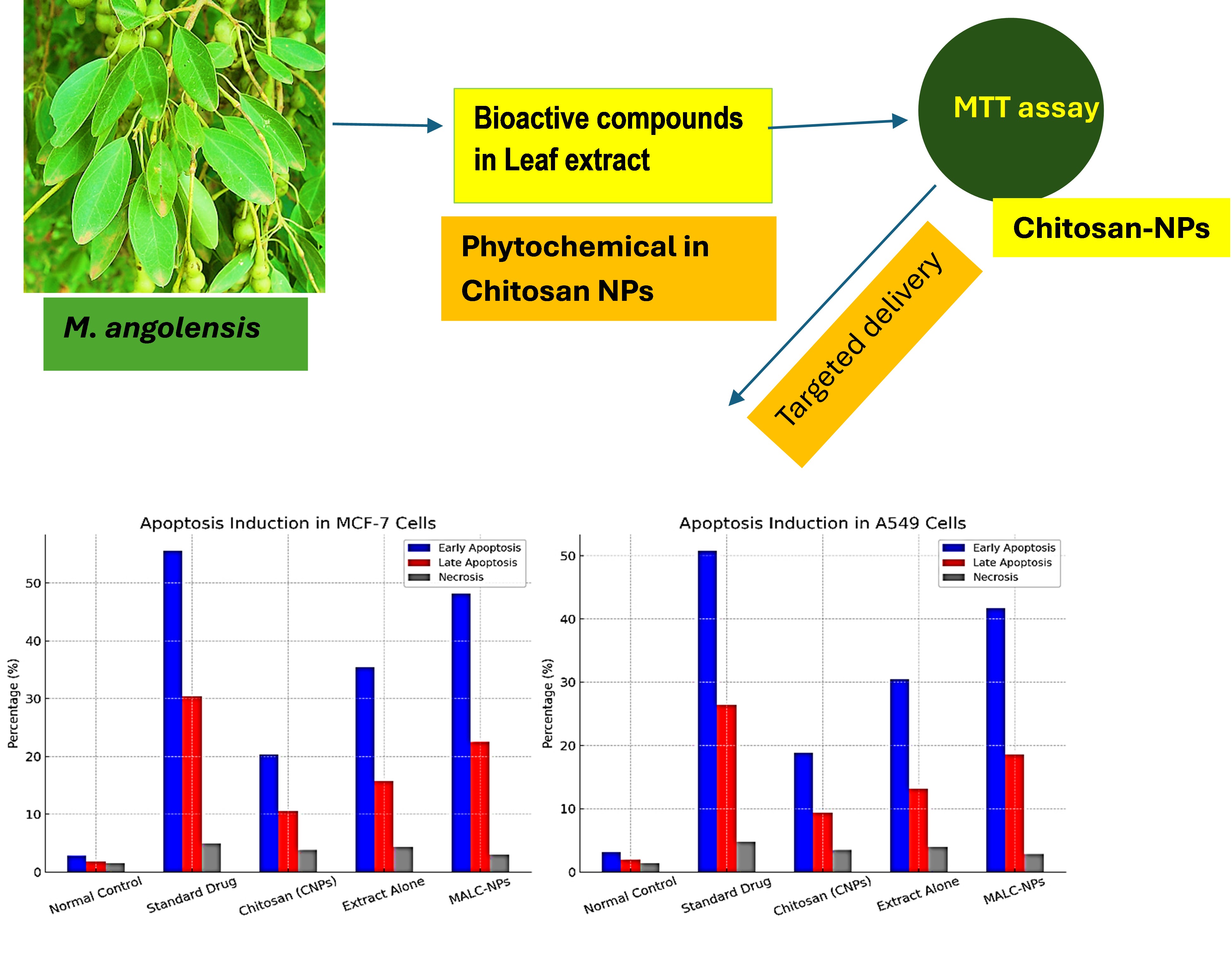
Methods: During the study, chitosan nanoparticles were fabricated using the ionic gelation method and later the M. angolensis leaves were encapsulated in them. The dynamic light scattering (DLS), Fourier-transform infrared (FTIR) spectroscopy, and scanning electron microscopy (SEM) techniques were used to assess the physicochemical properties of MALC-NPs. The effectiveness of the anti-inflammatory activity was analyzed by measuring the amount of nitric oxide (NO) produced and the activity of the cyclooxygenase-2 (COX-2) enzyme was measured. The MCF-7 (breast cancer) and A549 (lung cancer) cell lines were used to determine the cytotoxicity of MALC-NPs by MTT assay, while flow cytometry using Annexin V-FITC/PI staining reagents was performed to assess apoptosis induction.
Results: MALC-NPs showed particle size of 150 ± 5 nm, zeta potential of +32.6mV, encapsulation efficiency of 86.4%, which indicates the stability of the nanoparticle and effective loading of drug. In vitro assays showed greater anti-inflammatory activities, significant inhibition of NO production (IC₅₀: 21.5 µg/mL) and COX-2 activity (IC₅₀: 18.7 µg/mL) over time. The evaluation of cytotoxicity demonstrated the dose-dependent inhibition of MCF-7 (IC₅₀: 27.8 µg/mL) and A549 (IC₅₀: 32.4 µg/mL) carcinoma cells. Apoptosis induction was validated by flow cytometry. The early apoptosis values were 48.2% for MCF-7 and 41.7% for A549, late apoptosis values were 22.5 % and 18.6 %.
Conclusion: These results demonstrate the possible anti-inflammatory and anticancer actions of M. angolensis- chitosan nanoparticles. Their ability to inhibit inflammatory mediators suggest their possible use as an alternative therapeutic agent for inflammation-malignancy associated cancers. Further in vivo studies are required to confirm these actions and determine the mechanisms involved.
Disclaimer: This is not the final version of the article. Changes may occur when the manuscript is published in its final format.
We use cookies to improve your experience on our site. By continuing to use our site, you accept our use of cookies. Learn more